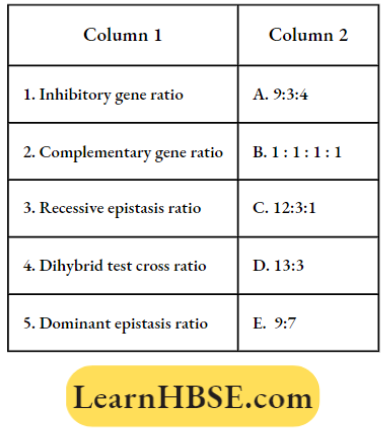
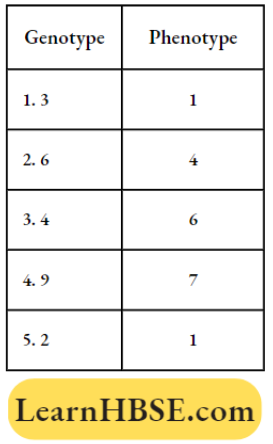
Oscillation And Waves Simple Harmonic Motion Long Answer Type Questions
Question 21. Show that the equation x = Acos(ωt- α) is a mathematical representation of simple harmonic motion. Find the time period and the maximum speed of the motion.
Asnwer:
Given equation
x = Acos(ωt-α)
∴ Velocity, v = \(\frac{d x}{d t}=-A \omega \sin (\omega t-\alpha)\)
The magnitude of sin (ωt-α) is maximum when it is ±1. This corresponds to the maximum velocity, or simply, the maximum speed.
∴ Maximum speed = ±Aω.
Again, cos (ωt-α) = cos(ωt- α + 2π) .
So, a particle executing SHM returns to its initial phase when the phase angle changes by 2π. The time elapsed between them is the time period (T) of the SHM.
∴ ω(t + T)-α = ωt-α + 2π
or, ωT = 2π or, T = \(\frac{2 \pi}{\omega}\)
Now, acceleration, a = \(\frac{d v}{d t}=-A \omega^2 \cos (\omega t-\alpha)\),
i.e., a = -ω²x
This relation signifies that the given equation is a mathematical representation of SHM.
Viscosity And Surface Tension Class 11 HBSE Notes
Question 22. If the frequency of a simple harmonic motion is n then find the frequency of its kinetic energy.
Answer:
Given that the frequency of a simple harmonic motion is n
If the particle executes the simple harmonic motion, then displacement, x = Asin(ωt+θ)
x = \(A \sin (\omega t+\theta)\)
= \(A \sin (2 \pi n t+\theta)\) (because \(n=\frac{\omega}{2 \pi}\))
∴ Velocity, v = \(\frac{dx}{dt}\) = Aω cos(ωt+θ)
So, the kinetic energy, if the particle has a mass m, is
K = \(\frac{1}{2} m v^2=\frac{1}{2} m A^2 \omega^2 \cos ^2(\omega t+\theta)\)
= \(\frac{1}{4} m A^2 \omega^2[1+\cos 2(\omega t+\theta)]\)
= \(\frac{1}{4} m A^2 \omega^2+\frac{1}{4} m A^2 \omega^2 \cos (2 \omega t+2 \theta)\)
On the right-hand side, the first term is a constant it ensures that the kinetic energy K is never negative. The cosine factor in the second term actually signifies that K is an oscillating quantity. The frequency of the oscillation is \(n^{\prime}=\frac{2 \omega}{2 \pi}=2 \cdot \frac{\omega}{2 \pi}=2 n .\)
The potential energy of an oscillating particle also oscillates with a frequency 2n.
Viscosity And Surface Tension Definition Class 11 Physics
Question 23. The kinetic energy of a particle is 1/2 mω²(A² -x²), where m, ω and A are constants. Prove that the motion of the particle is simple harmonic.
Answer:
Given
The kinetic energy of a particle is 1/2 mω²(A² -x²), where m, ω and A are constants.
Kinetic energy, K = \(\frac{1}{2} m v^2=\frac{1}{2} m \omega^2\left(A^2-x^2\right)\)
So, the velocity of the particle is,
v = \(\pm \omega \sqrt{A^2-x^2}= \pm \omega\left(A^2-x^2\right)^{1 / 2}\)
∴ \(\frac{d v}{d x}= \pm \omega \cdot \frac{1}{2}\left(A^2-x^2\right)^{-\frac{1}{2}} \cdot(-2 x)=\mp \frac{\omega x}{\sqrt{A^2-x^2}} .\)
Now, acceleration,
a = \(\frac{d v}{d t}=\frac{d x}{d t} \frac{d v}{d x}=v \frac{d v}{d x}\)
= \(\pm \omega \sqrt{A^2-x^2} \cdot \text { (Ғ) } \frac{\omega x}{\sqrt{A^2-x^2}}=-\omega^2 x .\)
So. a = -ω²xt which signifies that the motion is simple harmonic.
Numerical Problems On Viscosity And Surface Tension Class 11
Question 24. The speed of a body of mass m, when it is at a distance x from the origin, is v. Its total energy is \(\frac{1}{2} m v^2+\frac{1}{2} k x^2\), where k is constant. Prove that the body executes a simple harmonic motion.
Answer:
Given
The speed of a body of mass m, when it is at a distance x from the origin, is v. Its total energy is \(\frac{1}{2} m v^2+\frac{1}{2} k x^2\), where k is constant.
Total energy, E = \(\frac{1}{2} m v^2+\frac{1}{2} k x^2\)
∴ \(\frac{d E}{d t}=\frac{1}{2} m \frac{d}{d t}\left(v^2\right)+\frac{1}{2} k \frac{d}{d t}\left(x^2\right)\)
= \(\frac{1}{2} m \frac{d}{d v}\left(v^2\right) \frac{d v}{d t}+\frac{1}{2} k \frac{d}{d x}\left(x^2\right) \frac{d x}{d t}\)
= \(\frac{1}{2} m \cdot 2 v \cdot a+\frac{1}{2} k \cdot 2 x \cdot v\)
= \(m v a+k x v \quad[a=\text { acceleration }]\)
If E = constant, \(\frac{d E}{d t}=0\).
∴ \(m v a+k x v=0 \text { or, } a=-\frac{k}{m} x=-\omega^2 x\left[\omega= \pm \sqrt{\frac{k}{m}}\right]\)
This is the equation of a simple harmonic motion.
Viscosity And Surface Tension Derivation Class 11 HBSE
Question 25. The mass and radius of a satellite is twice that of the Earth. If a second pendulum is taken to that satellite, what will be Its time period?
Answer:
Given
The mass and radius of a satellite is twice that of the Earth. If a second pendulum is taken to that satellite
Acceleration due to gravity on the earth’s surface, g = \(\frac{GM}{R^2}\)
Acceleration due to gravity on the surface of the satellite in question, \(g^{\prime}=\frac{G \cdot 2 M}{(2 R)^2}=\frac{1}{2} \frac{G M}{R^2}\)
∴ \(\frac{g}{g^{\prime}}=2\)
Time period on the surface of the earth T = \(2 \pi \sqrt{\frac{L}{g}}\)
The time period on the surface of that satellite \(T^{\prime}=2 \pi \sqrt{\frac{L}{g^{\prime}}}\)
Hence, \(\frac{T}{T^{\prime}}=\sqrt{\frac{g}{g}}\)
or, \(T^{\prime}=T \times \sqrt{\frac{g}{g^{\prime}}}=2 \sqrt{2}=2.828 \mathrm{~s} \quad(because T=2 \mathrm{~s}) .\)
Factors Affecting Viscosity And Surface Tension Class 11
Question 26. A lift is moving vertically upwards with an acceleration a. What will be the changed time period of a simple pendulum suspended from the roof of the lift? If the lift becomes free and starts falling down with the acceleration due to gravity, what will be the change in the time period?
Answer:
Given
A lift is moving vertically upwards with an acceleration a.
Acceleration due to gravity (downwards) = g
Acceleration of the lift (upwards) = -a
Hence, the effective acceleration due to gravity with respect to the lift, g’ = g- (-a) = g + a
Therefore, the time period of the pendulum in the lift, \(T^{\prime}=2 \pi \sqrt{\frac{L}{g+a}}\)
We compare it with the time period inside the lift with no acceleration: \(T=2 \pi \sqrt{\frac{L}{g}}\)
∴ \(\frac{T}{T^{\prime}}=\sqrt{\frac{g+a}{g}} \text { or, } T^{\prime}=T \sqrt{\frac{g}{g+a}}\)
As g< (g+ a), T’ <T; hence, in this case, the time period will decrease. When the lift is falling freely downwards, its acceleration a = g.
Hence, the acceleration due to gravity with respect to the lift, g’ = g- a = g- g = 0.
Hence, the time period, in this case, T’ = \(2 \pi \sqrt{\frac{L}{g^{\prime}}}\) = infinity.
An infinite time period signifies that the pendulum will not oscillate at all.
Difference Between Viscosity And Surface Tension Class 11
Question 27. The bob of a simple pendulum is hollow. If half of it is filled with water, what will be the effect on Its time period?
Answer:
Given
The bob of a simple pendulum is hollow. If half of it is filled with water
The center of gravity of the hollow bob is at its center. Thus, the distance from the point of suspension to the center of the bob is the effective length (L) of the pendulum. When the bob is half filled with water, the center of gravity of the bob is lowered. Thus, the effective length of the pendulum increases. Hence, the time period (T) of oscillator of the pendulum also increases as T ∝ √L.
Question 28. A hollow sphere is filled up with water and is suspended using a long string. As water trickles out slowly through an orifice at the lower part of the sphere, it is observed that the time period of the pendulum first increases and. later continues to decrease. Explain these observations.
Answer:
Given
A hollow sphere is filled up with water and is suspended using a long string. As water trickles out slowly through an orifice at the lower part of the sphere, it is observed that the time period of the pendulum first increases and. later continues to decrease.
- Time period of a simple pendulum at a fixed place is directly proportional to the square root of the effective length of the pendulum. The distance between the point of suspension and the center of gravity of the bob is the effective length. The time period of oscillation increases or decreases with the increase or decrease of its effective length.
- The Centre of gravity of the spherical bob, completely filled with water, lies at the center of the sphere. As water trickles out through the orifice, the upper part of the sphere starts getting emptied making the lower half heavier than the upper half.
- As a result, the center of gravity of the bob starts going down from the center of the sphere and the effective length begins to increase. The time period also starts increasing.
- When more water trickles out, the center of gravity of the bob again starts shifting towards the center of the sphere resulting in a decrease in the effective length, and consequently, a lowering of the time period.
When the hollow sphere becomes completely empty, the center of gravity shifts back to the center of the sphere and the effective length regains its initial value. As a result, the final value of the time period equals its initial value.
Viscosity And Surface Tension Formula Class 11 HBSE
Question 29. The bob of a simple pendulum is made of iron. Exactly below the equilibrium position of the pendulum, the pole of a strong magnet is placed. What will be the change in the time period of the pendulum?
Answer:
Given
The bob of a simple pendulum is made of iron. Exactly below the equilibrium position of the pendulum, the pole of a strong magnet is placed.
In addition to the gravitational force, the magnetic force of attraction of the magnet acts on the pendulum bob. If acceleration produced by the magnetic field on the bob is a, the total acceleration of the pendulum bob g’ = g+a
∴ Time period of the simple pendulum T = \(x2 \pi \sqrt{\frac{L}{g+a}}\)…(1)
It is understood from equation (1) that the time period decreases due to the increase in acceleration of the pendulum bob.
Question 30. If the point of suspension of a simple pendulum is in a horizontal motion, with constant acceleration, what will be the effect on the time period?
Answer:
Given
The point of suspension of a simple pendulum is in a horizontal motion, with constant acceleration
When the point of suspension moves horizontally with a constant acceleration a, the effective acceleration due to gravity with respect to the point of suspension, \(g^{\prime}=\sqrt{g^2+a^2}\)
∴ Time period, T = \(2 \pi \sqrt{\frac{L}{\left(g^2+a^2\right)^{1 / 2}}}\)…(1)
From equation (1), it is understood that the time period decreases due to the increase in the value of the acceleration of the bob.
Question 31. A simple pendulum, suspended from the roof of a car, is oscillating. What will be the time period of the pendulum when the car moves in a circular path at a constant speed?
Answer:
Given
A simple pendulum, suspended from the roof of a car, is oscillating.
Two forces act on the pendulum bob when the car moves in a circular path at constant speed:
- Weight of the bob mg vertically downwards,
- Centrifugal force in the bob mω²r horizontally. (M=mass of bob, ω angular velocity of bob, r=radius of the circular path)
∴ Resultant effective force on the bob, F = \(\sqrt{m^2 g^2+m^2 \omega^4 r^2}=m \sqrt{g^2+\omega^4 r^2}\)
Hence, acceleration of the bob a = \(\frac{F}{m}=\sqrt{g^2+\omega^4 r^2}\)
∴ Time period T = \(2 \pi \sqrt{\frac{L}{a}}=2 \pi \sqrt{\frac{L}{\left(g^2+\omega^4 r^2\right)^{1 / 2}}}\)
Applications Of Viscosity And Surface Tension Class 11
Alternative Method: When the car along with the point of suspension of the pen¬dulum moves on a circular path horizontally, a freely falling body (along the vertical line through the point of suspension) is subjected to centripetal acceleration horizontally and acceleration clue to gravity vertically. The centripetal acceleration is ω²r (ω= angular velocity of point of suspension, r = radius of the circular path).
∴ Acceleration of the body falling freely with respect point of suspension = \(\sqrt{g^2+\omega^4 r^2}\)
Hence, time period of oscillation of the pendulum T = \(2 \pi \sqrt{\frac{L}{\left(g^2+\omega^4 r^2\right)^{1 / 2}}}\)
Experiment On Viscosity And Surface Tension Class 11 HBSE
Question 32. State the changes in the time period of a pendulum, when
- The pendulum is taken to a mountaintop from the earth’s surface
- The pendulum is set on the floor of a mine
- The diameter of the bob of the pendulum is increased
- Keeping the radius of the bob unchanged, its mass is increased. Give reasons for your answer
Answer:
Time period of a simple pendulum T = \(2 \pi \sqrt{\frac{L}{g}}\)
- Taking the pendulum from the earth’s surface or a mountain top, the value of g decreases. Hence, the time period increases.
- On the floor of a mine, the value of g decreases. Hence, the time period of a pendulum increases.
- On increasing the diameter of the bob, its effective length increases. Hence, the time period increases
- Time period is independent of the mass of the pendulum bob. Hence, even if mass is increased, keeping the diameter the same, the time period remains unchanged.